Canine CDV Antigen Pet Rapid Test&Equipment Dog Of Distemper Rapid Test
Product Details:
| Place of Origin: | China |
| Brand Name: | v-chek |
| Certification: | CE |
| Model Number: | DC001 |
Payment & Shipping Terms:
| Minimum Order Quantity: | 10000test |
|---|---|
| Price: | usd3usd/test |
| Packaging Details: | 10test/box |
| Delivery Time: | 7days |
| Payment Terms: | T/T, Western Union |
| Supply Ability: | 3500000test/day |
|
Detail Information |
|||
| Product Name: | Canine Distemper Virus Antigen | Use: | Vet Diagnosis |
|---|---|---|---|
| Sensitivity: | 99.8% | Cetification: | ISO13485 CE |
| Specimen: | Blood | Application: | Home Hospital |
| Highlight: | Canine Pet Rapid Test&Equipment,Distemper Pet Rapid Test&Equipment,Canine distemper rapid test |
||
Product Description
Canine CDV Distemper Antigen Rapid Test Kits dog of distemper Rapid Test Kit
Intended Use
The canine distemper virus antigen(CDV-Ag)Rapid Quantitative Test is a fluorescence immunoassay used along with CVF200 for quantitative determination of CDV-Ag concentration in canine nasopharyngeal or ocular secretion specimen. The test is used as an aid in the diagnosis and monitoring of canine distemper virus infection.
For in vitro diagnostic use only. For professional use only.
![]()
![]()
![]()
| Product name: Pet virus detection test kit |
| Applicable dog breeds: general for dogs |
| Validity period: one and a half years |
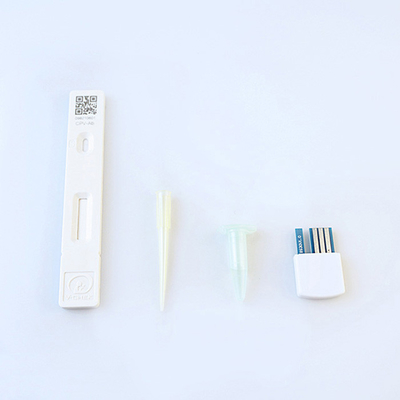
| Product packaging: box packaging,25bags/box, a manual |
| Product configuration: one swab,a set of test cards,a bottle of diluent |
1.This kit is for in vitro diagnostic use only. Do not swallow.
2.Lot Number of all the test components (test device, ID chip and buffer) must match each other. Do not mix components from different kit lots.
3.Inspect the packaging and labels before use. Do not use if the pouch is broken, torn or not well sealed, or the vial looks damaged or leaked.
4.Carefully follow the instructions and procedures described in this insert.
5.Do not use the test device beyond the expiration date. The test device must remain in its original sealed pouch until ready to use. Do not use if the pouch or the device itself is damaged, torn or not fully sealed.
6.A buffer tube should be used for processing one sample only.
7.The operation shall be conducted away from vibration and magnetic field. CVF200 VET may generate minute vibration during use, which should be regarded as normal.
8.One pipette tip should be used for one specimen only.
9.Do not touch the test area of the test device.
10.All specimens and used test materials are considered as potentially infectious. The used pipette tips, buffer tubes, test devices and specimens must be handled carefully and disposed of in accordance with local regulations and procedures.
![]()
![]()
![]()
![]()
![]()
![]()
![]()